Are you struggling with achieving the right balance of flexibility and strength in your PVC products? Many manufacturers face this challenge when formulating soft PVC compounds1.
DOP (Dioctyl Phthalate)2 plasticizer3 significantly impacts soft PVC performance by reducing hardness, decreasing tensile strength4, and increasing elongation at break5. The molecular insertion mechanism6 between PVC chains determines these property changes, making DOP concentration critical for optimal product performance.
Understanding how DOP affects your PVC formulation can make the difference between a successful product and costly production issues. Let me share what I've learned from 15 years of PVC compound manufacturing.
What Makes DOP Such an Effective Plasticizer for PVC?
Many people ask me why DOP remains the most widely used plasticizer3 in PVC applications. The answer lies in its unique molecular behavior.
DOP functions as an external plasticizer3 through dipole interactions and steric hindrance7 effects with PVC molecules. This mechanism allows DOP molecules to insert between PVC polymer chains without altering the chemical structure of the base resin.
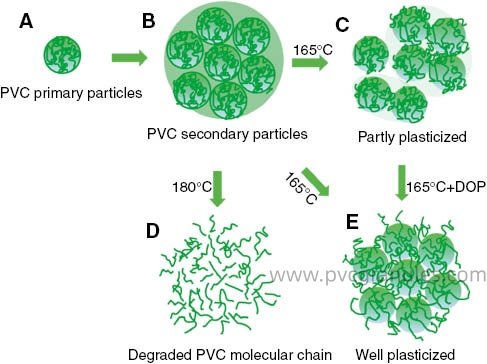
Plasticizers fall into two main categories: internal and external. Internal plasticizer3s chemically bond with the polymer structure during synthesis. External plasticizer3s like DOP work differently. They physically integrate into the polymer matrix through molecular forces.
The science behind DOP's effectiveness centers on molecular mobility8. When DOP molecules position themselves between PVC chains, they create space and reduce intermolecular forces9. This process weakens the van der Waals forces that normally restrict polymer chain movement.
I've observed this mechanism countless times in our laboratory. The dipole interactions between DOP's ester groups and PVC's chlorine atoms create stable associations. These interactions don't form chemical bonds, but they're strong enough to maintain plasticizer3 retention during processing and end-use.
| Property | Without DOP | With DOP | Change Mechanism |
|---|---|---|---|
| Chain Mobility | Restricted | Enhanced | Reduced intermolecular forces9 |
| Glass Transition | High Tg | Lower Tg | Increased free volume |
| Flexibility | Rigid | Flexible | Enhanced segmental motion |
The steric hindrance7 effect also plays a crucial role. DOP molecules occupy space between polymer chains, preventing tight packing. This spacing effect reduces crystallinity and promotes amorphous regions10 where polymer chains can move more freely.
How Does DOP Concentration Affect PVC Hardness Properties?
The relationship between DOP content and PVC hardness follows a predictable pattern that I've documented across thousands of formulations.
Shore hardness11 decreases proportionally as DOP plasticizer3 content increases. This occurs because DOP molecules insert between PVC chains, weakening intermolecular forces9 and reducing the energy required for segmental chain motion.
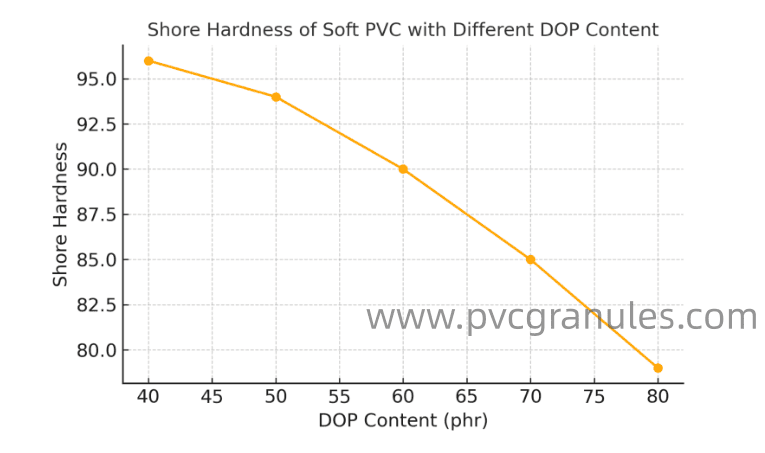
The hardness reduction mechanism involves multiple factors working together. Primary among these is the reduction of intermolecular attraction forces. PVC without plasticizer3 exhibits strong dipole-dipole interactions between polymer chains. These forces create a rigid structure with high resistance to deformation.
When we add DOP to PVC, the plasticizer3 molecules position themselves strategically between polymer chains. This insertion creates physical separation and reduces the strength of intermolecular forces9. The result is a softer material that deforms more easily under applied stress.
In our production facility, I've measured this relationship across various DOP concentrations. At 10 PHR (parts per hundred resin), we typically see Shore A hardness around 95-98. Increasing to 30 PHR drops hardness to 85-90 Shore A. At 50 PHR, hardness falls to 70-80 Shore A.
The energy barrier for molecular motion decreases significantly with DOP addition. Without plasticizer3, polymer chains require substantial energy to overcome intermolecular forces9 and move past each other. DOP reduces this energy barrier by creating lubrication12 between chains and providing free volume for movement.
| DOP Content (PHR) | Shore A Hardness | Chain Mobility | Typical Applications |
|---|---|---|---|
| 10-20 | 90-95 | Limited | Semi-rigid profiles |
| 30-40 | 80-90 | Moderate | Flexible tubing |
| 50-70 | 65-80 | High | Cable jacketing |
| 80+ | <65 | Very High | Gaskets, seals |
Temperature also influences this relationship. At elevated processing temperatures13, DOP molecules become more mobile and distribute more evenly throughout the PVC matrix. This improved distribution enhances the plasticizing effect and creates more uniform hardness properties.
Why Does DOP Reduce PVC Tensile Strength Performance?
The inverse relationship between DOP concentration and tensile strength4 represents one of the most significant trade-offs in PVC formulation.
Tensile strength decreases as DOP concentration increases because excessive plasticizer3 disrupts interfacial compatibility between PVC matrix components. This disruption damages interfacial bonding and compromises mechanical integrity of the polymer network.
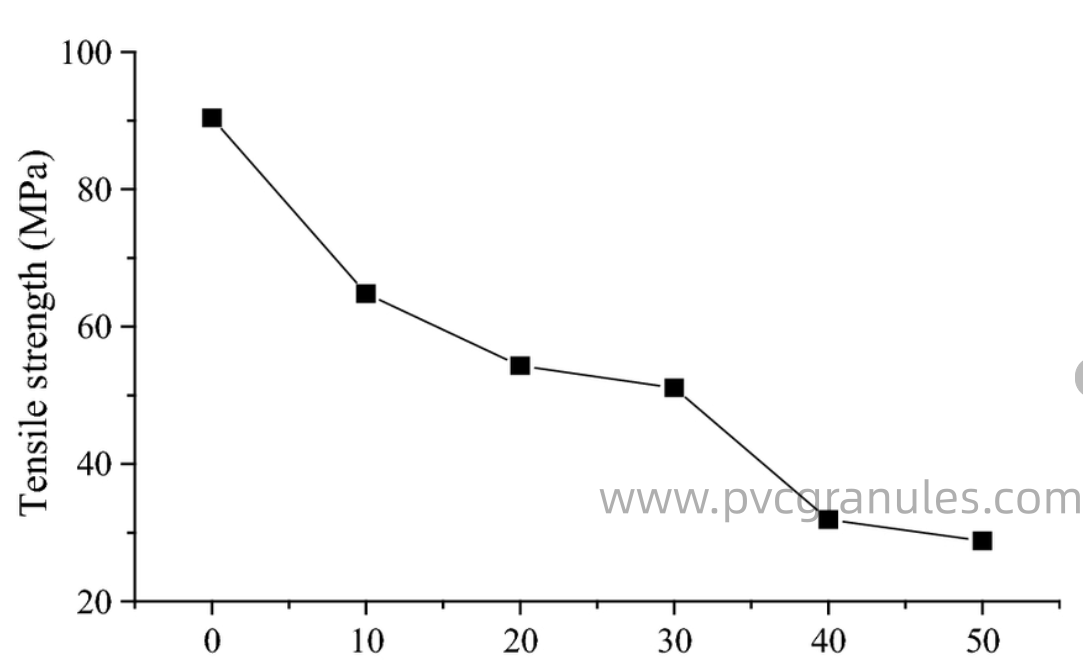
The mechanism behind this strength reduction involves several interconnected factors. First, DOP molecules create discontinuities in the polymer matrix. While these discontinuities enhance flexibility, they also create weak points where stress concentration occurs under tensile loading.
Interfacial compatibility becomes critical at higher DOP loadings. When plasticizer3 concentration exceeds the polymer's absorption capacity, phase separation14 begins. This separation creates domains of concentrated DOP that act as stress concentrators during mechanical testing.
I've documented this phenomenon extensively in our quality control laboratory. At low DOP concentrations (10-20 PHR), tensile strength4 remains relatively stable because the plasticizer3 integrates well within the PVC matrix. The polymer chains maintain sufficient interaction to transfer stress effectively.
As DOP content increases beyond 30-40 PHR, we observe significant strength degradation. The excess plasticizer3 begins to form separate phases or pools within the matrix. These plasticizer3-rich regions cannot contribute to load-bearing capacity and instead act as defects that promote crack initiation and propagation.
The molecular weight distribution15 of the PVC resin also influences this relationship. Higher molecular weight PVC shows better plasticizer3 compatibility and maintains strength at higher DOP loadings. Lower molecular weight resins reach saturation more quickly and exhibit earlier strength loss.
| DOP Level (PHR) | Tensile Strength (MPa) | Strength Retention (%) | Matrix Condition |
|---|---|---|---|
| 0 | 45-50 | 100 | Rigid, brittle |
| 20 | 35-40 | 80-85 | Good integration |
| 40 | 25-30 | 60-65 | Moderate separation |
| 60 | 15-20 | 35-45 | Significant phase separation14 |
| 80+ | 8-15 | 20-30 | Poor matrix integrity |
Processing conditions significantly affect this strength-plasticizer3 relationship. Proper mixing and temperature control help maximize DOP distribution and minimize phase separation14. Our twin-screw extrusion process maintains precise temperature profiles to optimize plasticizer3 incorporation while preserving mechanical properties.
How Does DOP Enhancement Improve PVC Elongation Properties?
The positive correlation between DOP content and elongation at break5 represents the primary benefit of plasticization in flexible PVC applications.
Elongation at break increases proportionally with DOP concentration because the plasticizer3 enhances polymer chain mobility, reduces crystallinity, and promotes plastic deformation under stress. This mechanism transforms brittle PVC into flexible, stretchable material.
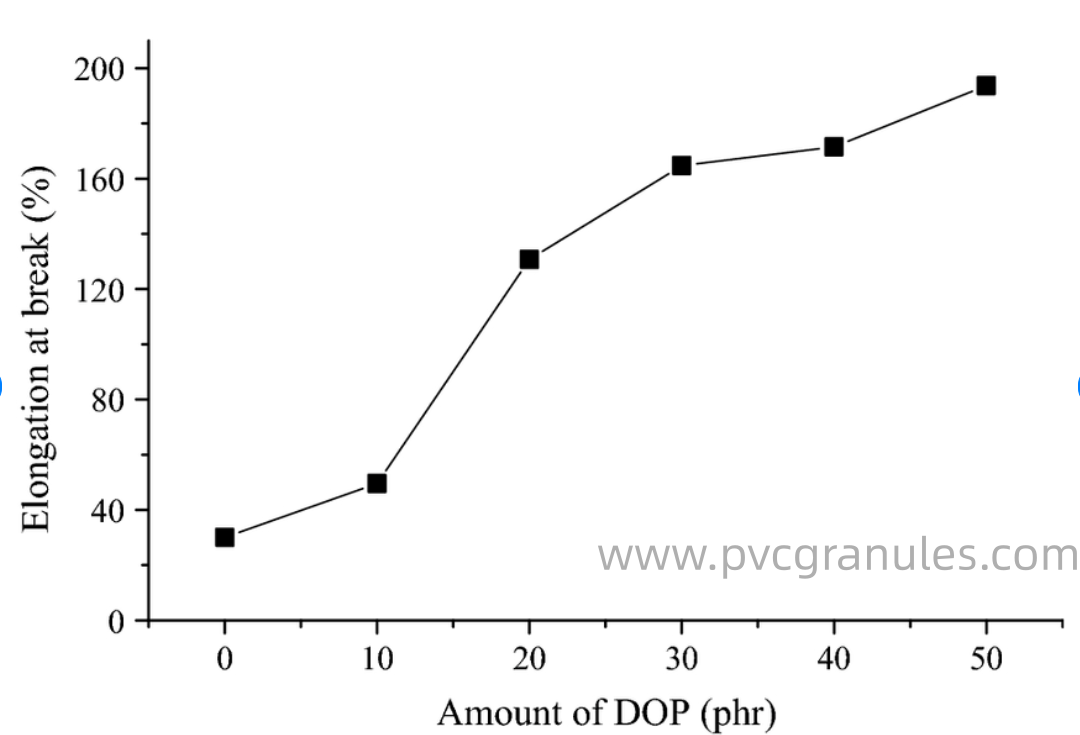
The elongation enhancement mechanism operates through multiple pathways. Primary among these is the increased segmental mobility of polymer chains. DOP molecules act as internal lubricants, allowing chain segments to slide past each other more easily during deformation.
Crystallinity reduction plays an equally important role. Unplasticized PVC contains crystalline regions where polymer chains pack tightly in ordered arrangements. These crystalline domains resist deformation and promote brittle failure. DOP disrupts this ordered packing and promotes amorphous regions10 where chains can move freely.
In our testing laboratory, I've measured elongation improvements across various DOP concentrations. Rigid PVC typically exhibits elongation values of 5-15%. Adding 30 PHR DOP increases elongation to 150-200%. At 50 PHR, elongation reaches 250-300%. Higher loadings can achieve elongations exceeding 400%.
The stress-strain behavior changes dramatically with DOP addition. Rigid PVC shows linear elastic behavior followed by brittle fracture. Plasticized PVC exhibits initial elastic deformation, followed by yield, then extensive plastic flow before ultimate failure.
Chain entanglement density affects elongation response. Higher molecular weight PVC resins with more entanglements show greater elongation improvements with plasticizer3 addition. The entanglements act as temporary crosslinks that prevent premature chain pullout during stretching.
| DOP Content (PHR) | Elongation at Break (%) | Deformation Mode | Typical Performance |
|---|---|---|---|
| 0-10 | 5-50 | Brittle fracture | Poor flexibility |
| 20-30 | 150-250 | Ductile yielding | Good flexibility |
| 40-50 | 250-350 | Extensive flow | Excellent flexibility |
| 60+ | 350-500+ | Rubber-like | Outstanding flexibility |
Temperature sensitivity of elongation properties varies with DOP concentration. Highly plasticized compounds maintain flexibility at lower temperatures compared to lightly plasticized materials. This temperature independence makes DOP-plasticized PVC suitable for outdoor applications16 with varying climate conditions.
The molecular structure of DOP contributes to its elongation-enhancing effectiveness. The branched octyl chains provide excellent compatibility with PVC while maintaining plasticizer3 efficiency. Linear plasticizer3s often show different elongation responses due to variations in molecular packing and chain interactions.
Conclusion
DOP plasticizer3 fundamentally transforms soft PVC performance through molecular-level interactions that reduce hardness, decrease tensile strength4, and dramatically improve elongation properties for enhanced flexibility.
Learn about soft PVC compounds and their significance in various industries. ↩
Explore the role of DOP in PVC formulations to understand its impact on flexibility and strength. ↩
Discover the function of plasticizers in enhancing the properties of polymers like PVC. ↩
Learn about tensile strength and its significance in evaluating material performance. ↩
Explore the concept of elongation at break and its importance in flexible materials. ↩
Understand the molecular interactions that influence the performance of PVC products. ↩
Discover how steric hindrance influences the behavior of molecules in materials. ↩
Learn about molecular mobility and its impact on the flexibility of polymer materials. ↩
Understand the role of intermolecular forces in determining the properties of polymers. ↩
Learn about the significance of amorphous regions in enhancing material flexibility. ↩
Get insights into Shore hardness and its relevance in material testing. ↩
Discover the role of lubrication in improving the processing and properties of polymers. ↩
Find out how temperature influences the properties and processing of PVC materials. ↩
Explore the phenomenon of phase separation and its effects on material properties. ↩
Understand the significance of molecular weight distribution in polymer performance. ↩
Understand the factors that affect PVC performance in outdoor environments. ↩
